Fares BASSIL
Multiple system atrophy: a translational approach. Characterization of the insulin/IGF-1 signaling pathway
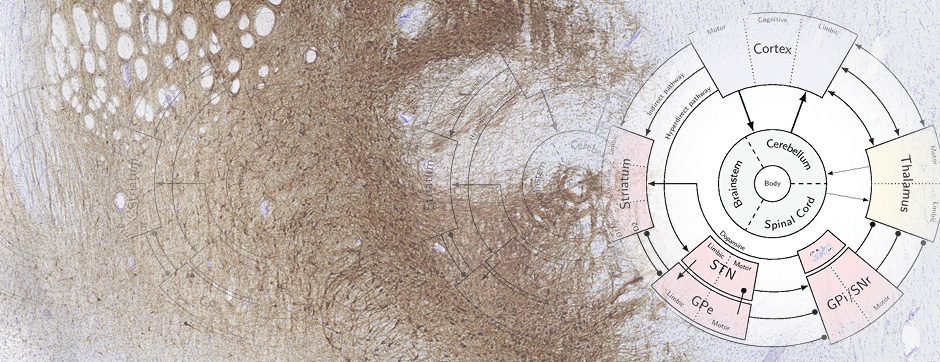
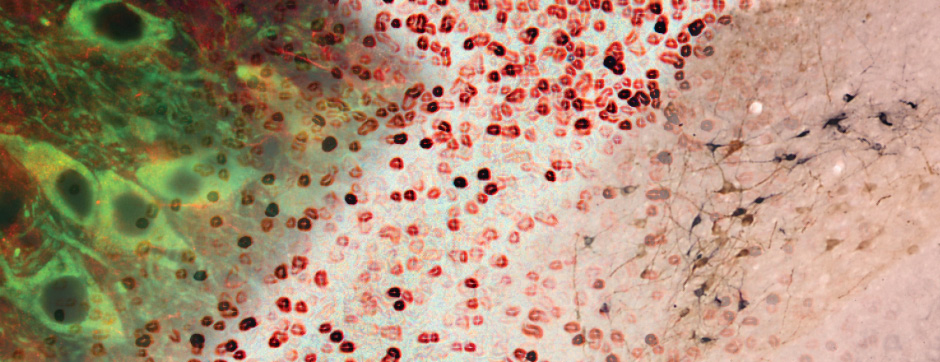
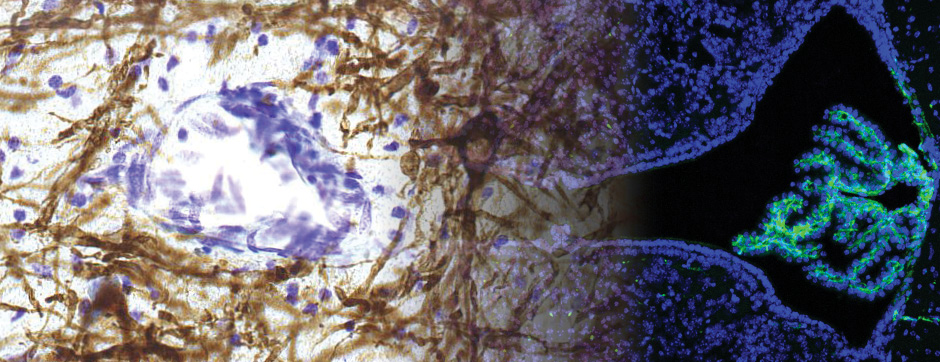
septembre 2015 Directeur(s) de thèse : Wassilios MEISSNER Résumé de thèseThis work focused on translational approaches in synucleinopathies and more specifically in multiple system atrophy (MSA). Beyond their role in glucose homeostasis, insulin/IGF-1 are neurotrophic factors in the brain. Studies have shown altered insulin/IGF-1 signalling in Alzheimer’s disease and data suggest impaired insulin signaling/IGF-1 in Parkinson’s disease (PD) and MSA. The aim of my work was to characterize insulin/IGF-1 signalling in MSA and PD brain tissue. Both groups showed neuronal insulin resistance. Oligodendrocytes in MSA patients were also insulin resistant.
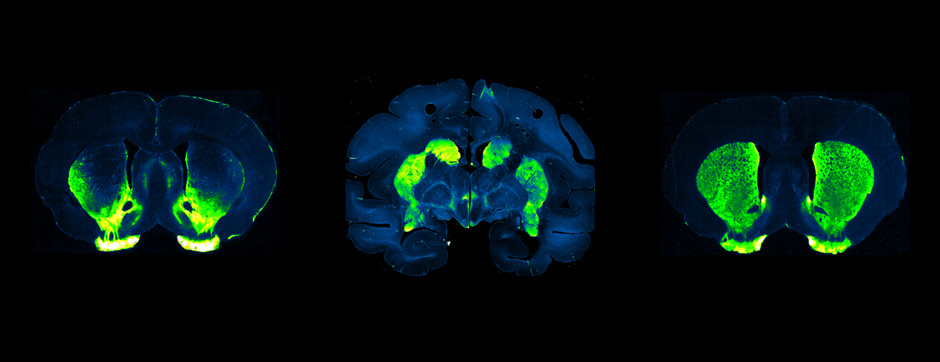
In line with the translational approach, we also targeted α-synuclein (α-syn) truncation pharmacologically in MSA transgenic mice, which led to reduced α-syn aggregation and the protection of dopaminergic neurons.
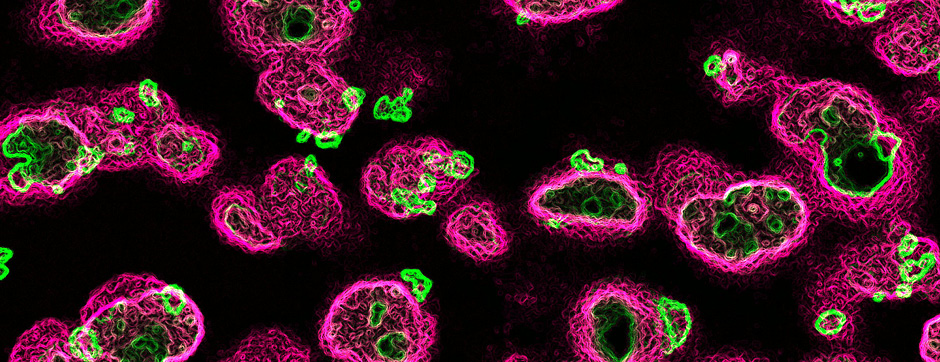
We also assessed the activity and distribution of matrix metalloproteinases (MMPs) in the brain of MSA patients compared to healthy controls. MMPs are involved in the remodelling of the extracellular matrix, demyelination, α-syn truncation and blood brain barrier permeability. We showed altered expression and activity of MMPs in two distinct structures in MSA brains. We were also able to show that glial cells were the source of increased MMPs and show a unique expression of MMPs in α-syn aggregates of MSA patients compared to PD, evidence that might hint at a mechanism that is differently altered between PD and MSA.
We here show distinct pathological features of MSA such as key alterations occurring in oligodendrocytes, further supporting MSA as a primary oligodendrogliopathy. We also present VX-765 as a candidate drug for disease modification in synucleinopathies.
Keywords: Synuclein, insulin resistance, matrix metalloproteinase, multiple system atrophy, Parkinson’s disease, insulin, insulin like growth factor-1, glucagon like peptide-1, glial cytoplasmic inclusions, postmortem human brain study, rodent, translational approach, c-terminal truncation.